Profit Server уже почти 10 лет работает в сфере предоставления услуг по аренде выделенных
Ищете хостинг или виртуальный сервер с комфортными условиями по оплате и использованию? Такие решения
Перечень операций в рамках настройки 1С и стоимость услуг зависит от специфики компании, внедряемого/настраиваемого
При заказе VPS/VDS от CloudVPS можно воспользоваться готовыми дистрибутивами для быстрого старта.
Ветровки изначально были созданы для спортсменов, чтобы защитить их от ветра и легкого дождя
JUSTHOST — провайдер услуг, у которого есть все необходимое для уверенного присутствия в интернете.
Согласно статистике ежегодно российские маркетплейсы растут x2. Это касается, как заработка самих площадок, так
Зимой уборка снега с крыш промышленными альпинистами в Москве очень необходима для безопасности. Такую
Октябрь - 2022 | Лучшая бюджетная мышь для игр является важной частью вашей недорогой
⭐⭐⭐⭐⭐Лучшие беспроводные мыши: рейтинг мышек 2020 года, их основные отличия. ✅На что стоит обратить
Обзор и сравнение лучших игровых мышек для компьютера из разных ценовых категорий - до
Рейтинг лучших цветных МФУ для дома по мнению экспертов и отзывам покупателей. Номинации рейтинга:
Лучшие лазерные принтеры для дома 2022 года по мнению пользователей. Качественные и недорогие модели
Какой принтер выбрать для дома по соотношению цена качество и по отзывам покупателей? Представляются
Давайте подробно рассмотрим 5 лучших мониторов с частотой 144 Гц, доступных в настоящее время.
Обзор и описание мониторов 24 дюйма. Рейтинг ТОП-12 лучших моделей 2020-2021 года по соотношению
Лучшие бюджетные мониторы: топ 2022 года по мнению покупателей. Краткий обзор моделей в ценовом
Мы составили для вас рейтинг ТОП-10 лучших моделей графических планшетов для рисования на 2021-2022
В рейтинге представлены 10 лучших планетарных миксеров 2021-2022 года. Все их объединяет отличное качество,
Рейтинг лучших планшетов по отзывам покупателей. Какой планшетный компьютер лучше выбрать: диагональ, ОС, производительность
Обзор и рейтинг лучших клавиатур мембранного типа по отзывам покупателей и независимым оценкам экспертов.
Рейтинг лучших клавиатур для компьютера с подсветкой. Как выбрать игровую клавиатуру с подсветкой клавиш?
Топ лучших ноутбуков для работы и учебы 2021 года. Актуальный рейтинг ноутбуков 2021 года
Рейтинг лучших модемов для интернета. В обзоре представлены самые удачные модели модемов для интернета
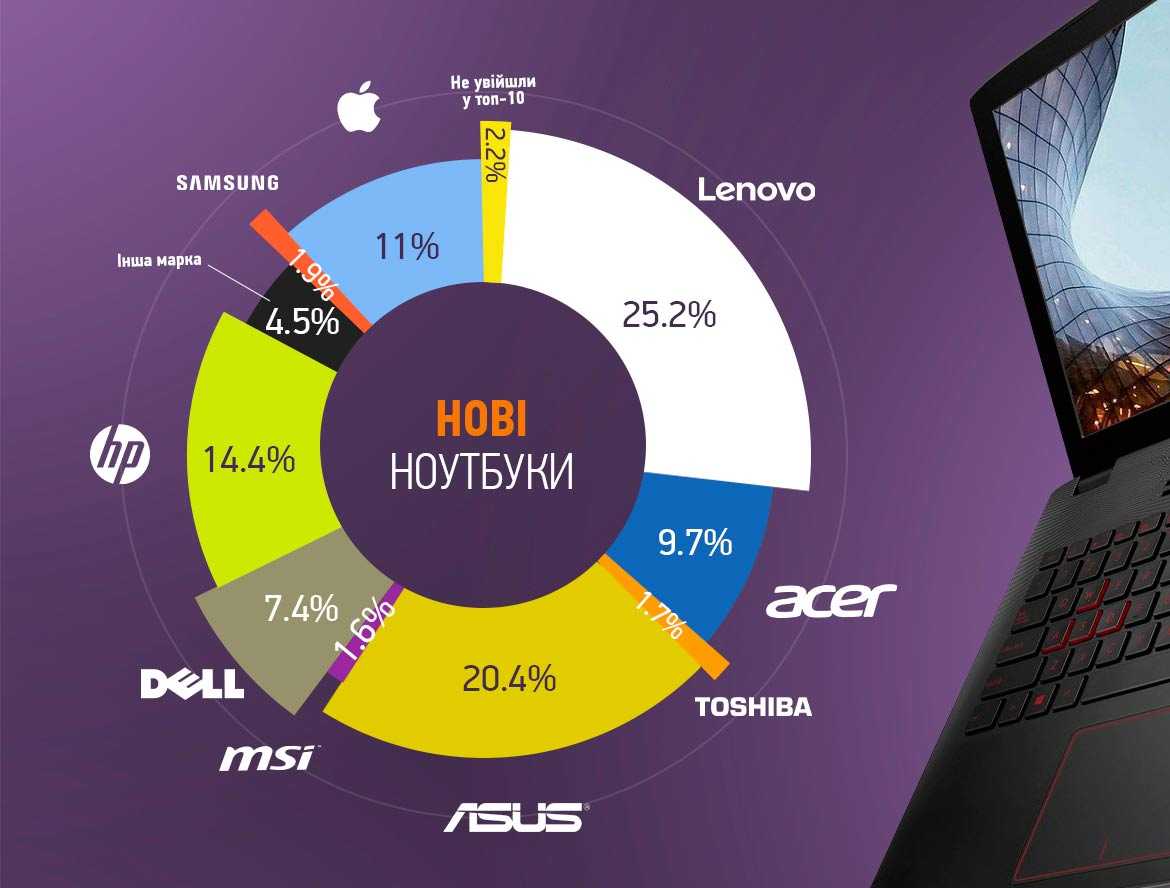
Прочитав наш рейтинг, вы будете знать, ноутбук какого производителя лучше купить в 2022 году.
Какой Макбук лучше купить в 2021 году? Полный и подробный разбор всех актуальных моделей